Activities
ISIUM
ISIUM 2023 conference «Improving Use of Medicines: Connecting, Learning, Moving Forward», October 28-30, 2023, Chiang Mai, Thailand.
EPN
The Ecumenical Pharmaceutical Network (EPN) is a Christian not-for-profit organization, with a global network of over 118 members in 37 countries.

Antimicrobial Resistance
ReACT - ReAct is one of the first international independent networks to articulate the complex nature of antibiotic resistance and its drivers.
ISDB
The International Society of Drug Bulletins (ISDB) is a world wide network of bulletins and journals on drugs and therapeutics that are financially and intellectually independent of pharmaceutical industry. It was founded in 1986, with the support of the WHO Regional Office for Europe. The rationale for the Society is that drug bulletins independent of funding from industry experience problems not faced by editors and publishers of other journals.
HAI
At Health Action International, we are dedicated to advancing policies that enable all people to realise the human right to the highest attainable level of health. To make this a reality, our team and global network carry out research to inform evidence-based advocacy for equitable access to affordable medicines and healthcare.
Rational Use of Medicines - Semey, Kazakhstan
Резолюция по рациональному применению лекарственных средств принята в г. Семей, Казахстан, 8 декабря, 2017 года




III Congress of Clinical Pharmacologists of Kazakhstan. 12-13.10.2023
III Congress of Clinical Pharmacologists of Kazakhstan
with international participation, dedicated to the 70th
Anniversary of the State Prize laureate of the Republic
of Kazakhstan, Doctor of Medical Sciences, Professor Alexander Evgenievich Gulyaev.
The Congress is organized by the following universities: WKMU (West Kazakhstan Marat Ospanov Medical University — Department of Pharmacology), AMU (Astana Medical University — Department of Clinical Pharmacology), KMU (Karaganda Medical University — Departments of Clinical Pharmacology and Evidence-Based Medicine), SMU (Semey Medical University — Department of Pharmacology named after Doctor of Medical Sciences, Professor M.N. Musin), SKMA (South Kazakhstan Medical Academy — Department of Pharmacology, Pharmacotherapy, and Clinical Pharmacology), POCP (Professional Association of Clinical Pharmacologists and Pharmacists), CoRSUM — Coalition for Rational and Safe Use of Medicines.
Congress dates: 12.10.23-13.10.23
Organizers: Ministry of Health of the Republic of Kazakhstan, Marat Ospanov WKMU, POCP (Professional Association of Clinical Pharmacologists and Pharmacists), CoRSUM — Coalition for Rational and Safe Use of Medicines. Chairman of the Organizing Committee: Chairman of the Board-Rector of Marat Ospanov WKMU, Doctor of Medical Sciences, Professor A.A. Kaliyev.
The congress covered the following directions:
- Continuous Integrated Medical Education (CIME) in the teaching of Clinical
- Ensuring Medication Safety.
- Issues of Antibiotic Therapy and Antibiotic Resistance.
- Rational Use of Medications.
- Electronic Information Resources to Assist Clinical Pharmacologists and Pharmacists.
- Experimental Pharmacology: Opportunities and Perspectives.
- Young Researchers; Competition.

More information about III Congress of Clinical Pharmacologist of Kazakhstan in MEDEX_September_2023
… responding to current health care needs and trends
The role of the pharmacist in promoting the rational use of drugs has been recognised and discussed for many decades in Russian-speaking countries and pharmaceutical care is now undergoing unprecedented changes to include clinical pharmacy practice.
The development of this new specialty in the Commonwealth of Independent States countries was the focus for a major session during the Congress of Pharmacologists and Clinical Pharmacists of Kazakhstan held on April 5, 2022. The conference – The Third International Scientific and Practical Conference: Rational Use of Innovative Medicines in Practical Medicine – was held in Almaty and was attended by specialists from Kazakhstan, Kyrgyzstan, Ukraine, Moldova, Russia, Great Britain, Armenia, and New Zealand.
The practice of clinical pharmacy is well recognised and established in developed countries but is still in its infancy in developing countries, including many Russian-speaking countries.
Over the last decade there have been many workshops, conferences and training sessions held in Kazakhstan to highlight the importance of including clinical pharmacy in the State register of specialties. Some initial steps have been taken but the development of the specialty is still in its early stages and the involvement of pharmacists in direct patient care is limited.
The current pharmacy curriculum is inadequate. It provides limited didactic and practical exposure and does not include a clinical component. There is a mismatch between learning objectives and course content, there are different approaches to examinations, there is a shortage of expert lecturers and there is no regulatory framework. Misconceptions among health professionals about the role of clinical pharmacist are prevalent.
To produce well-trained and competent pharmacists who can provide direct clinical patient care, pharmacy educators need to strengthen and develop the
pharmacy curriculum.
Some of the recommendations for this include:■ teaching practical skills,
■ establishing models of practice in pharmacy,
■ incorporating the practical experience gained in academic settings into existing clinical initiatives,
■ implementing clinically based interventions during ward rounds,
■ adapting and implementing models such as problem-based learning and ‘pharmacy curriculum development and validation’.
It is important to understand that if clinical pharmacy is to survive and grow, it must gain acceptance by medical professionals and the community.
Moreover, more effort is required to expand and improve the clinical pharmacy program so that its benefits can be reaped by the local people.
The participants of the conference emphasised the importance and necessity of studying programs offered by leading international educational institutions for implementation in the curriculum of Kazakhstan.
Международный Конгресс «GLOBAL HEALTH», Алматы, Казахстан. 23.09.2022
23 сентября 2022 года в Алматы состоялся VII Международный Конгресс «Global Health», посвящённый 25-летию основания Казахстанского медицинского университета «ВШОЗ» и подписания Соглашения между Европейским Региональным Бюро ВОЗ и Министерством здравоохранения Республики Казахстан
Члены CoRSUM Проректор Медицинского Университета Елена Леонидовна Степкина и Президент CoRSUM Наталья Александровна Чеботаренко участвовали в работе секции «Фармация».


More information about Congress «Global Health», Almaty, Kazakhstan, WHOZ. MEDEX_October_2022.
Клинические навыки фармацевтов: подход, ориентированный на пациента.
Clinical Skills for Pharmacists. A Patient — Focus Approach.
II Congress of Clinical Pharmacologists of Kazakhstan. Semey Medical University, March, 24-25, 2022
II Congress of Clinical Pharmacologists of Kazakhstan dedicated to the memory of Professor Dr. Talgat Mazhitov with international participation. The conference is held on the initiative of the following universities: NCJSC «Medical University of Semey» (Department of Pharmacology named after M.N. Mussin), NCJSC «Medical University Astana» (Department of Clinical Pharmacology), Astana «Medical University Karaganda» (Departments of Clinical Pharmacology and Evidential Medicine), NCJSC «West Kazakhstan Marat Ospanov Medical University» (Department of Pharmacology), JSC «South Kazakhstan Medical Academy», CoRSUM «Coalition for Rational and Safe Use of Medicines», Location:Republic of Kazakhstan, Semey, Nur-Sultan. Congress dates: March 24-25, 2022.
Organizers: Ministry of Health of the Republic of Kazakhstan, NCJSC «Semey Medical University», NCJSC «Astana Medical University», CoRSUM «Coalition for Rational and Safe Use of Medicines». Chairman of Organizing Committee: Head of Department of Pharmacology named after Doctor of Medical Sciences, Professor M.N. Musin, NCJSC «Semey Medical University», Candidate of Medical Sciences, Tuleutayeva R.Ye.
Deputy Chairman of the Organizing Committee: Head of the Department of Clinical Pharmacology «Astana Medical University» Dr. M.S., Akhmadyar N.S.

More information about II Congress of Clinical Pharmacologist of Kazakhstan in MEDEX_05_2022.
The work of the congress hold in the following areas:
- Training of clinical pharmacology in medical universities of Kazakhstan: new directions and prospects.
- Rational use of drugs and issues of personalized pharmacotherapy in clinical medicine.
- Antimicrobial chemotherapy drugs.
- New approaches to the treatment of Covid-19 in adults: a review of international guidelines.
- Young scientists competition.



The work of the congress was carried out in 5 directions:
1. Problems of teaching clinical pharmacology at medical universities of Kazakhstan.
2. Questions of the rational and personalized pharmacotherapy in clinical medicine.
3. Chemotherapy drugs.
4. Pharmacotherapy and prevention of COVID-19.
5. Young scientists competition
Organizers:
Ministry of Health of the Republic of Kazakhstan,
NJSC «Astana Medical University», NJSC «KAZNMU named after S.D. Asfendiyarov «.
The congress hold on May 27-28, 2021 by the initiative of the Department of Clinical Pharmacology of the Astana Medical University, the Department of Clinical Pharmacology and Evidence-Based Medicine of the Medical University of Karaganda, the Department of Pharmacology of the Semey Medical University, Department of Clinical Pharmacology KAZNMU “S.D. Asfendiyarov», Department of Pharmacology, Department of General and Clinical Pharmacology KazMNO, Almaty, CoRSUM -Coalition on Rational and Safe Use of Medicines.
Dates and venue: Kazakhstan, Nur-Sultan, Almaty.

Head of the Department of Clinical Pharmacology «Astana Medical University» Dr. M.S., Akhmadyar N.S.
More information about I Congress of Clinical Pharmacologist of Kazakhstan in MEDEX _June_2021_en
WHO Good Governance for Medicines programme: an innovative approach to prevent corruption in the pharmaceutical sector.
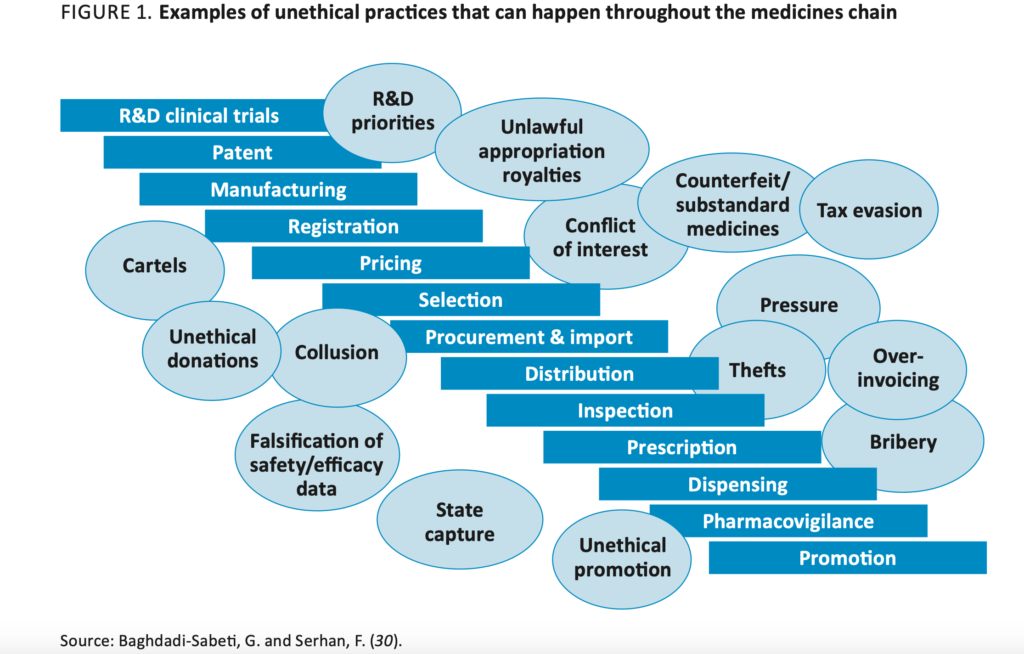
- Ministry of Health (e.g. health service department, pharmaceutical units, national programmes for disease control, medical stores, procurement division);
- Medicine Regulatory Authority (registration, inspection, control of promotion, licensing and clinical trial units/departments, etc.);
- Procurement agencies, importers and distributors both from the private and the public sector (including tertiary-care hospitals, primary‐care facilities, pharmaceutical brokers and consolida- tors, hospital pharmacists);
- Members of committees, such as tender committees, therapeutics committees, selection of essen- tial medicines committees, at national and local level;
- Ministries of finance, industry and commerce, customs and importation;
- Local anti-corruption organizations/commission;
- National quality control laboratories;
- Audit departments (internal, external, and state auditors);
- Pharmaceutical industry (multinational and national) and associations;
- Nongovernmental organizations, such as those engaged in health service activities, patient advo-
- cacy groups, “watch‐dog” organizations;
- International donor organizations, such as the World Health Organization, the United Nations
- Children’s Fund, the United Nations Development Programme, the World Bank and the Global
- Fund;
- Academic institutions (national colleges, state universities and research institutes);
- Professional associations (medical, pharmacy, biochemist associations, etc.);
- Media (if knowledgeable about the pharmaceutical sector);
- Ethics committees, institutional review boards;
- Health insurance funds.
The resolution of the conference Millennium Development Goal's Implementation in NIS Countries. CoRSUM and Association of Pharmacists of Moldova in collaboration with WCC, EPN, Scientific Centre of Drug and Medical Technology Expertise of Armenia, Lviv State Medical University and Public Health School of Kazakhstan. Введите текст заголовка
World Pneumonia Day in Moldova, November, 12, 2011
Participants discussed the Millennium Development goals’ implementation in context of public health issues. The main challenges in public health in Moldova and other NIS countries were highlighted as priorities:
- Lack of modern AMR surveillance system in the public health system, including lack of AMR Hospital Passport.
- Low level of effectiveness of microbiology laboratory service and discordance between microbiological service and public health hospitals.
- Lack of coordination between human health system and veterinary / agriculture systems in AMR issues.
- Insufficient educational programs in medical school and lack of awareness campaign among population in AMR.
- Urgent need for National AMR strategic plan and National AMR Task group.
The Conference adopted the Declaration on Antimicrobial resistance that was initially developed during European AMR day – November, 18 and later widely discussed by all CoRSUM members.
- Lack of pediatrics dosages and formulations is a barrier to effective, qualitative medical care of children as in Moldova as well in other NIS countries. The main challenge is a absence of necessary medicines to treat HIV/AIDS and Tuberculoses. In spite of the growing rate of TB and AIDS in children during last 6 years, there is the gap in the procurement /ordering of medicines for children.
- In spite of the fact that the majority of anti-Tb drugs and ARVs are procure through GFTM Fund and hopefully are prequalified by WHO Prequalification program, some of anti-Tb drugs are produced in Moldova. There is a concern about the quality of such medicines.
- The lack of independent/ unbiased sources of literature about medicines exists in Moldova still. General practitioners and pharmacists do not have access to evidence –based information as about medicines as well about treatment protocol s.
- There is a insufficient level of transparence in the drug registration and drug regulation in general processes. Existing processes of the registration of medicines as well as procedures of clinical trials and decision making process about the registration are not transparent.
- The educational programs in high school do not use the modern electonical sources of information such Pubmed, Cochrain, NICE, BNF and others.
- Many public health specialists do not use computer, internet , CD or other sources of information. Post -graduation education does not provide the possibility to obtain new skills in computer/internet still.
- In spite of the adopted National Medicine Policy (MOH RM №1352-XV from 03.10.2003: 4.4; 4,6) the Professional Associations and Non-for- profit, non-governmental organizations working in public health are not involved in the formulation, adaptation and discussion of the decision process about the current medicine policy.
- Pharmaceutical and informational support to the patient with socially-significant diseases (especially diabetes) appears in incomplete volume.
- In Moldova there is the absence of the control system on the promotion of the food supplements to consumers and accompanying, correct, corresponding to the law on protection of the consumer’s rights information.
The Conference calls to:
- To develop concrete, detailed program on AMR with all stakeholders as from governmental organizations as well from non-governmental, professional medical, pharmaceutical organizations, agriculture/ veterinary structures and Church-related institutions.
- In the ordering medicines to take into account the amount of pediatrics dosages and formulations for children , especially with social diseases such as tuberculosis, HIV/AIDS.
- International Organizations that assist countries with medicines supply such as Global fund for TB. AIDS and Malaria , UNICEF to take into account and to include in the plan of drug supply pediatrics medicines.
- To activate the work in the computerization of the pharmaceutical service, especially in the social important diseases such as diabetes, TB and other.
- Standard treatment Guidelines develop and adopt with wide range of medical and pharmacy specialists.
- To determine conceptual new ways of the collaboration between physicians, pharmacists and other specialists of public health.
- To continue the collaboration between governmental and non-governmental organizations in the rational and safe use of medicines in the framework of partnership of CoRSUM.